Methane has been getting much bad press lately. Despite all of the bad things projected onto methane there are many aspects and uses of this natural gas making it valuable and important. Using methane as a fuel for electricity and heat generation and feedstock for U.S. chemical and refining processes has resulted in an unprecedented amount of abundance for much of the world today. Methane will continue to help power prosperity in the world for the foreseeable future.
This blog topic discusses some of the properties, sources, uses and usage of methane today. Also covered are some of the aspects of methane as an air pollutant when it is released to the atmosphere.
Figure 1. Tetrahedral projection of methane molecule.
Chemistry
- A methane molecule is made of one atom of carbon with four atoms of hydrogen with the chemical formula of CH4. The “C” stands for carbon; the “H” stands for hydrogen.
- Methane has a tetrahedral molecular geometry with a carbon atom at the center and four hydrogen atoms making up the corners of the tetrahedron.
- It has a molecular weight of 16.04 pounds per pound mole.
- Methane’s gross heating value is approximately 1010 BTU per standard cubic feet (SCF).
- Methane is a compressible, colorless, odorless gas at ambient atmospheric conditions.
- As a gas it is flammable over a range of concentrations approximately 5 to 15% in air.
Sources of Methane
- Methane is a major component of natural gas produced at an oil and gas well production facility.
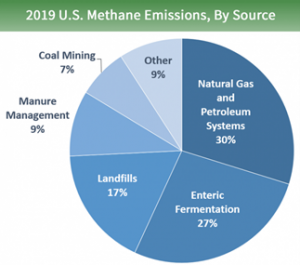
- Coal mining results in methane production. Targeted production of methane from coal beds is an ongoing source of methane production.
- Methane is found beneath permafrost and ocean sediments and is known as methane hydrate, methane clathrate and hydromethane, methane ice.
- Methane is produced by ruminant livestock (cattle, sheep, buffalo, and goats) as a byproduct of the microbes partaking in the digestive process. Ruminants belch methane. This is also called enteric fermentation. According to the EPA, cattle in the U.S. emit into the atmosphere approx. 5.5 million metric tons of methane per year (6.1 U.S. tons per year).
- Other significant sources of methane include landfills, manure management and other miscellaneous sources.
- A breakdown of methane emissions to the atmosphere by source type in the U.S published by the USEPA:
Uses and Usage
- Methane is used primarily as a clean fuel throughout the economies of the world. According to the U.S. Energy Information Agency (EIA), “In 2013, the United States consumed approximately 26.13 trillion cubic feet (Tcf) of natural gas.” The International Energy Agency (IEA) estimated that worldwide natural gas used the world used 3,500 billion cubic meters (123.6 Tcf) in 2013.
- The natural gas that local gas distribution utilities send to our homes is composed of primarily methane. Also, the natural gas will contain small percentages of ethane, propane and butane.
- Since methane is odorless, utilities add odorants chemicals called mercaptans to ensure people can detect a gas leak via the “rotten egg” smell of mercaptans.
- Technologies are being refined to convert methane to methanol. Being a liquid at atmospheric conditions, methanol can be stored and transported easier than methane gas. Methanol has many uses in the economy including use as a hydrate inhibitor in natural gas handling and processing.
- Large scale systems are used and continue to be refined for gas-to-liquids (GTL) technologies to make liquid fuels and products from natural gas and methane.
- Various industries and use methane in their processes
- A power plant using natural gas fuel generates approximately 13 kWh of electricity generated per cubic foot of natural gas (mostly methane) burned.
- Homeowners use methane for cooking and heating their homes.
Air Pollution
- Methane is regulated as a greenhouse gas (GHG) by the USEPA and has a global warming potential (GWP) of 25 (based on a 100 year horizon). By definition, carbon dioxide (CO2) has a GWP = 1. The higher the GWP for a greenhouse gas, the higher its “heat trapping” capacity by the pollutant. You determine the CO2e by multiplying the mass of a GHG by its GWP factor. This means that if a facility vents to the atmosphere, 100 tons of methane, then the CO2e (carbon dioxide equivalent) for the methane emissions are:
(25 lbs CO2e/lb CH4) * 100 tons = 2500 tons of CO2e.
- The combustion of methane results in 117 pounds of Carbon Dioxide (CO2) gas per MMBTU combusted.
- Since methane in not considered, by regulation, a significant photochemically reactive gas, it is not defined as a volatile organic compound (VOC).
- Incomplete combustion of methane can yield formaldehyde (formula: H2C=O). Interesting fact: formaldehyde is used to make formalin – the aqueous solution of formaldehyde that is used as embalming fluid.
- The USEPA has several programs to help companies maximize the amount of methane recovered from production processes and minimize the venting to the atmosphere. Some of these programs include Natural Gas STAR Program, Landfill Methane Outreach Program (LMOP), Coalbed Methane Outreach Program (CMOP)
Future
An Executive Order on Tackling the Climate Crisis at Home and Abroad was published online by the White House in January 2021. This executive order included efforts to reduce methane emissions from the oil and gas industry.
In November 2021, the USEPA posted online prepublication proposed rules to reduce methane emissions from existing and new oil and natural gas facilities. Cimarron will review this proposed regulation and publish a blog summarizing the potential impacts to the oil and gas industry.
Cimarron – Who We Are
Cimarron’s vision is to work with our clients to create a cleaner environment.
The company engineers and manufactures environmental, production and process equipment for the upstream, midstream and downstream energy industries, as well as environmental control solutions for biogas at wastewater facilities, digester tanks and landfills.
Cimarron offers our customers the know-how and environmental expertise to meet the environmental standards of today and tomorrow. Cimarron is committed to bring value to the Energy industry and their shareholders based on our financial strength, experienced personnel, and engineering capabilities.
As a company, we thrive every day to make a difference through innovation (e.g. ESG), customer focus, and operational efficiency. In addition to being present in all major regions in the US, Cimarron serves more than 45 countries around the world, ranging from offshore to desert. From key operational centers in the United States, Italy and the United Arab Emirates, Cimarron offers ongoing service and support through its own field service personnel and strategic third-party partners, creating a cleaner environment for our customers and their shareholders.
Since its founding in the mid-1970’s in Oklahoma, the company’s product offering has expanded from production equipment to include the largest line of environmental solutions that capture or incinerate fugitive vapors. With the acquisitions of HY-BON/EDI in 2019 and AEREON (including Jordan Technologies) in 2020, Cimarron has added strong brands, products, and services to its portfolio.
Please contact us to learn more about our products and services and about all our ESG solutions at sales@cimarron.com or visit our website cimarron.com.


